 3 illustrates this and other electron-pair geometries that minimize the repulsions among regions of high electron density (bonds and/or lone pairs). In molecules with more than three atoms, there are many more possible geometries. 2: The BeF 2 molecule adopts a linear structure in which the two bonds are as far apart as possible, on opposite sides of the Be atom. Each bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. Use this number to determine the electron pair geometry. Thus, the electron-pair geometry is tetrahedral and the molecular structure is bent with an angle slightly less than 109.5°. Step 3: Add these two numbers together to get the regions of electron density around the central atom. If we look at the nitrite ion NO2-, we have 2 Bond Pairs and 1 Lone pair of electrons. The Lewis structure of H 2 O indicates that there are four regions of high electron density around the oxygen atom: two lone pairs and two chemical bonds: Figure 7.6.9 7.6. The commercial name for methanol is wood alcohol. CH 3 OH is the chemical formula for methanol or methyl alcohol, a colorless sweet smelling, extremely volatile alcohol. In NO2, we have 2 Bond Pairs and 1 lone electron. Methanol (CH3OH) Lewis dot structure, molecular geometry or shape, electron geometry, bond angle, hybridization, formal charges, polar vs non-polar. The oxygen is bonded to 2 groups and has 2 lone pairs of electrons. 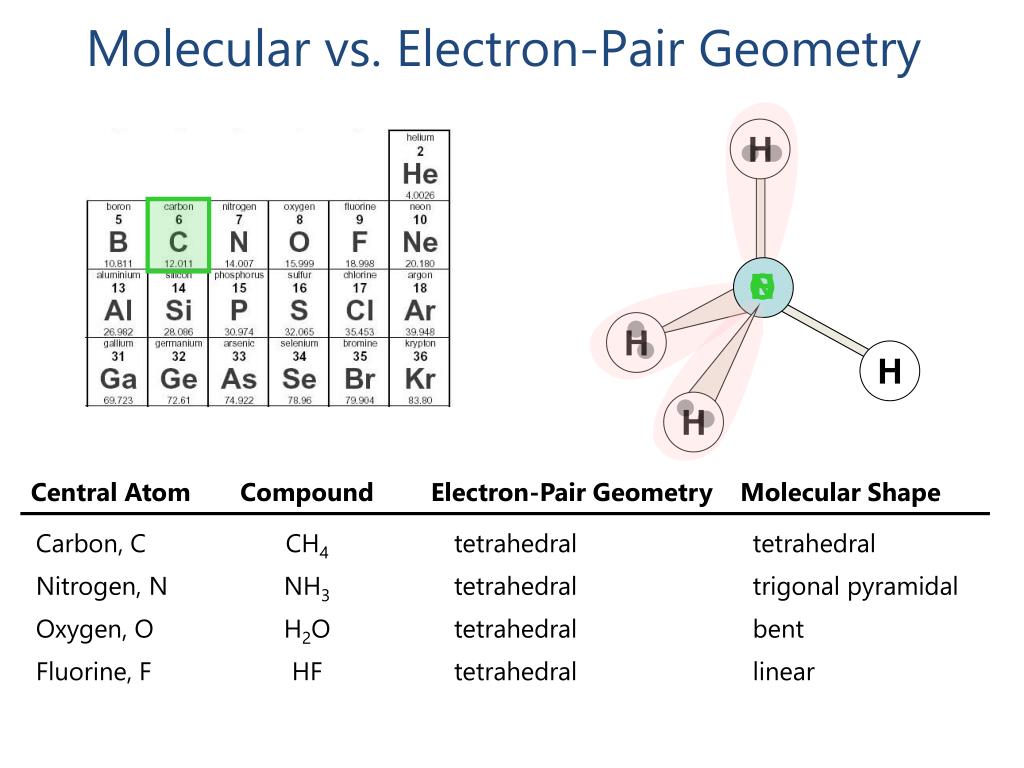
However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. VSEPR chart: We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees. The electron pair geometry and molecular geometry is trigonal planar with bond angles of 120 o. 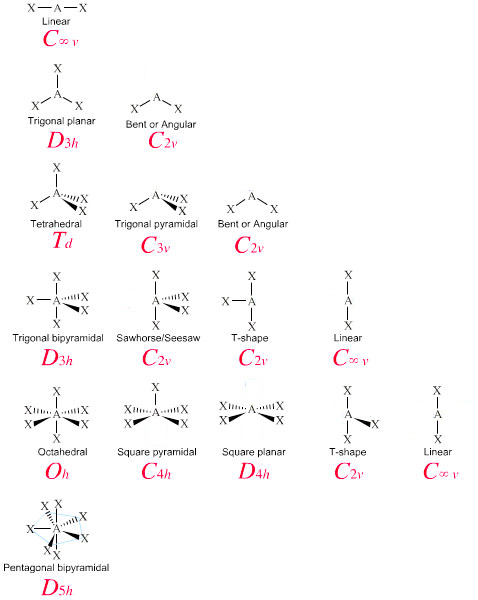
An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. A polyatomic molecule contains more than two atoms. An explanation of the molecular geometry for the XeF5 + ion (Xenon pentafluroide anion) including a description of the XeF5 + bond angles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
